Hydrolyzed Egg Membrane Market Forecast 2026–2036: Global Market to Reach USD 675.0 Million by 2036 at 12.9% CAGR
Clinical validation and regulatory clarity are accelerating adoption of hydrolyzed egg membrane in joint, beauty, and pet nutrition applications worldwide.
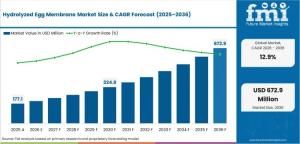
NEWARK, DE, UNITED STATES, March 4, 2026 /EINPresswire.com/ -- The global hydrolyzed egg membrane market is entering a decisive growth phase, driven by clinical validation, regulatory approvals, and next-generation joint formulations. According to the latest analysis by Future Market Insights (FMI), the market crossed USD 180.0 million in 2025 and is projected to reach USD 200.0 million in 2026. Over the forecast period, revenues are expected to surge to USD 675.0 million by 2036, expanding at a CAGR of 12.9%.
This represents an absolute dollar opportunity of USD 475.0 million between 2026 and 2036, signaling a structural shift from commodity collagen peptides toward specialized, bio-inductive membrane extracts. Procurement shortlists are increasingly narrowing toward clinically backed and traceable ingredients as enterprise buyers seek measurable efficacy.
Explore trends before investing – request a sample report today! https://www.futuremarketinsights.com/reports/sample/rep-gb-32152
Hydrolyzed egg membrane ingredients are derived from avian eggshell membranes using controlled enzymatic hydrolysis. These bioactive extracts are standardized for collagen, hyaluronic acid, and glycosaminoglycan (GAG) content and are primarily utilized in joint health, mobility, nutricosmetic, and animal nutrition applications.
Market Momentum Anchored in Clinical Substitution
Joint health & mobility supplements command 44% of total market revenue in 2026. Aging populations and rising preventive healthcare awareness are accelerating adoption beyond traditional osteoarthritis use cases. Clinical studies increasingly position hydrolyzed egg membrane as a low-dose alternative to bulky glucosamine or collagen regimens, improving patient compliance.
Regulatory clarity is reinforcing confidence. In 2025, a U.S. FDA notice established a daily background membrane intake benchmark of 46 mg/day, strengthening baseline safety metrics for novel food approvals. Additionally, recent EFSA evaluations confirmed a 2,000 mg/kg bw/day NOEL for membrane hydrolysates, raising the ingredient’s regulatory credibility across Europe.
FMI analysts highlight that documented efficacy has reset procurement behavior. A 2025 GRAS submission estimated an all-person mean intake of 308 mg/day for proposed food applications, underscoring safe scalability.
Powder Formats Lead with 52% Share
By form, powder and flakes account for 52% of ingredient demand in 2026. Their dominance stems from:
• Low moisture content and improved shelf stability
• Ease of incorporation into capsules, gummies, and functional foods
• Reduced freight and storage risks
• Standardized bulk packaging efficiencies
Advanced enzymatic hydrolysis technologies now deliver glycosaminoglycan yields up to 44.60 μg/mg, preserving peptide integrity while improving cellular absorption rates. Controlled 2025 trials demonstrated that 450 mg/day supplementation significantly improved hair density metrics among aging consumers, further expanding nutricosmetic appeal.
Geographic Growth Corridors
Country-level growth reflects regulatory readiness and aging demographics:
• United States: 13.1% CAGR
• China: 12.5% CAGR
• India: 12.2% CAGR
• Germany: 11.7% CAGR
• United Kingdom: 11.4% CAGR
• Brazil: 11.2% CAGR
North America leads global innovation due to stringent safety frameworks and extensive GRAS pathways. In 2025, post-marketing surveillance data recorded an exceptionally low adverse event rate of 2.16 per 1,000,000 servings, reinforcing ingredient reliability.
In Europe, compliance with EFSA documentation standards and non-GMO traceability mandates defines competitive positioning. Germany and the United Kingdom remain critical procurement hubs for clinically substantiated nutricosmetics.
Asia-Pacific is emerging as a high-volume opportunity zone. China’s expanding middle class and India’s growing awareness of bioavailable proteins are tightening the link between domestic wellness spending and imported ingredient adoption.
Supply-Side Realignment and Strategic Partnerships
Raw material volatility remains a structural restraint. The hydrolyzed egg membrane industry depends on commercial egg-breaking operations for feedstock supply. USDA data reported a 4% decline in total 2025 egg production to 105 billion units, intensifying procurement strategies focused on long-term feedstock contracts.
Market participants are responding with vertical integration and facility expansions. In September 2025, Stratum Nutrition opened a high-capacity production facility in Carthage, Missouri to scale extraction capabilities.
Industry partnerships are also strengthening distribution networks. As noted by Md Abul Hasnat, CEO of Alchem Enterprise, a new collaboration with Stratum Nutrition aligns marketing and regulatory expertise to accelerate global penetration of science-backed nutraceutical ingredients.
Meanwhile, in November 2026, Stratum Nutrition’s clinically validated OVOLUX™ ingredient secured the 2025 “Collagen Ingredient of the Year” award, recognizing advancements in sustainable beauty-from-within formulations.
Expanding Opportunity Matrix
Beyond joint support, hydrolyzed egg membrane is capturing adjacent revenue streams:
• Veterinary expansion: Premium pet food brands integrate human-grade extracts into joint-support treats amid rising pet humanization trends.
• Nutricosmetic convergence: Beauty brands combine structural proteins with vitamins to justify premium positioning.
• Geographic penetration: Localized warehousing and distribution partnerships reduce cross-border friction across Asia and Latin America.
FMI analysts observe that double-blind 2025 studies confirmed 300 mg/day of fresh membrane extracts outperformed 8,000 mg of hydrolyzed collagen in improving skin mechanical parameters, validating premium pricing models.
Competitive Landscape
Key players in the hydrolyzed egg membrane market include:
• Stratum Nutrition
• Biova LLC
• Eggnovo SL
• Kewpie Corporation
• Ecovatec Solutions
• Certified Nutraceuticals Inc.
Regulatory compliance, proprietary extraction technologies, and clinical substantiation remain decisive competitive barriers. Established suppliers leverage GRAS notifications, EFSA approvals, and independent toxicology assessments to secure long-term procurement contracts.
Analyst Perspective
Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, comments that the market is transitioning from novelty-driven adoption toward mechanism-based positioning. Long-term expansion will depend on quantifiable outcomes such as mobility improvements, stiffness reduction, and recovery timelines. Suppliers combining standardized extraction with transparent clinical communication will secure durable differentiation.
As regulatory thresholds rise and safety benchmarks tighten, hydrolyzed egg membrane is poised to evolve from a niche nutraceutical ingredient into a core regenerative formulation component across global mobility-health portfolios.
Why FMI: https://www.futuremarketinsights.com/why-fmi
Explore FMI's related ongoing Coverage in Food and Beverage Domain:
Fertility Supplement Market: https://www.futuremarketinsights.com/reports/fertility-supplements-market
Hydrolyzed Vegetable Protein Market: https://www.futuremarketinsights.com/reports/hydrolysed-vegetable-protein-market
Pet Food Ingredients Market: https://www.futuremarketinsights.com/reports/pet-food-ingredients-market
Agricultural Adjuvant Market: https://www.futuremarketinsights.com/reports/agriculture-adjuvants-market
Cereal Bars Market: https://www.futuremarketinsights.com/reports/cereal-bars-market
Sudip Saha
Future Market Insights Inc.
+ +1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.